Advanced Head and Neck Squamous Cell Carcinoma (HNSCC) or Neoadjuvant Followed by Adjuvant Therapy for Resectable Locally Advanced HNSCC
KEYTRUDA and KEYTRUDA QLEX™ (pembrolizumab and berahyaluronidase alfa-pmph) Injection 165 mg + 2,000 units/mL are each indicated for the treatment of adult patients with resectable locally advanced head and neck squamous cell carcinoma (HNSCC) whose tumors express PD-L1 [Combined Positive Score (CPS) ≥1] as determined by an FDA-authorized test, as a single agent as neoadjuvant treatment, continued as adjuvant treatment in combination with radiotherapy (RT) with or without cisplatin and then as a single agent.
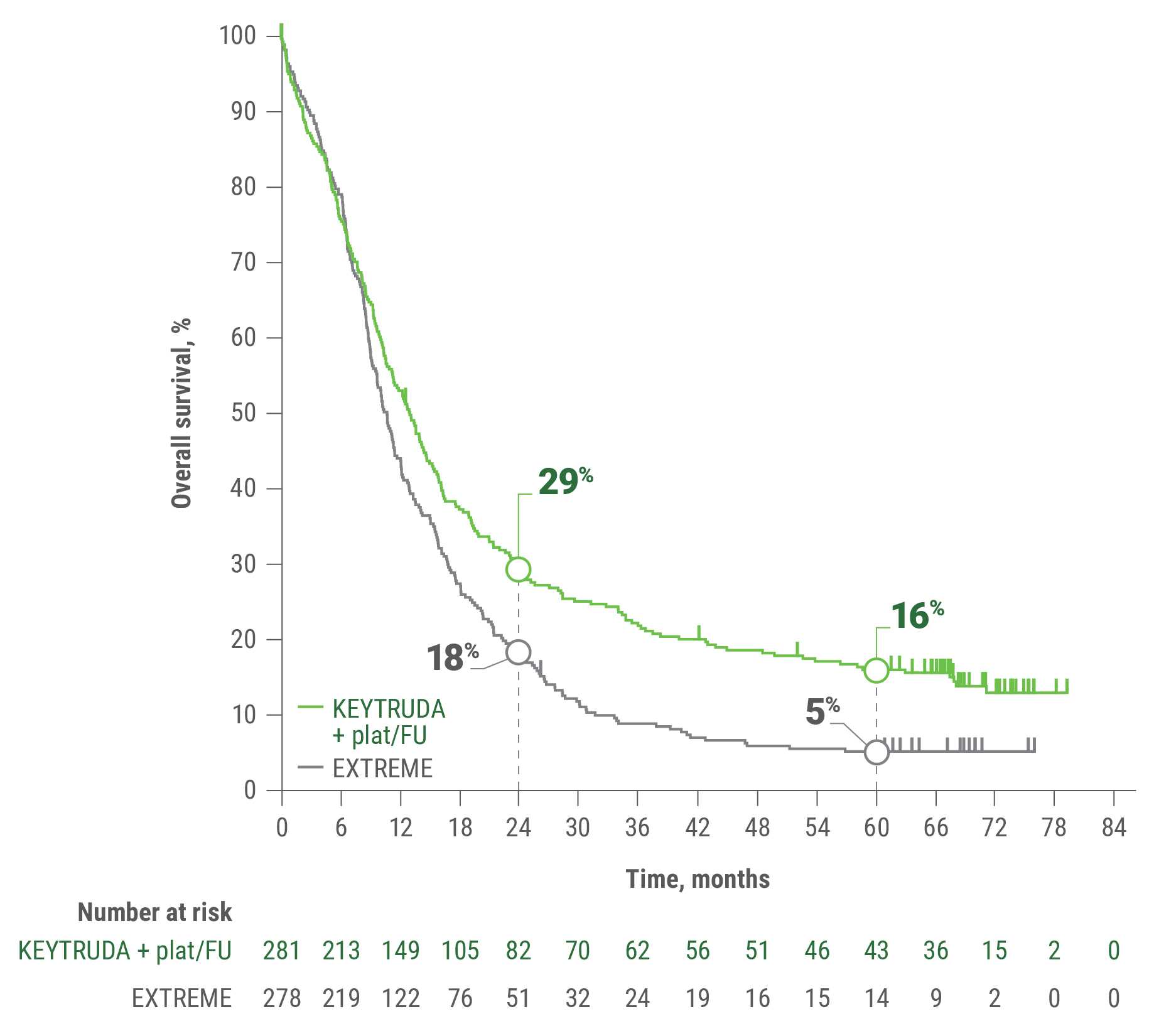
KEYTRUDA and KEYTRUDA QLEX™ (pembrolizumab and berahyaluronidase alfa-pmph) Injection 165 mg + 2,000 units/mL are each indicated, in combination with platinum and fluorouracil (FU), for the first-line treatment of adult patients with metastatic or with unresectable, recurrent head and neck squamous cell carcinoma (HNSCC).
KEYTRUDA and KEYTRUDA QLEX™ (pembrolizumab and berahyaluronidase alfa-pmph) Injection 165 mg + 2,000 units/mL, as single agents, are each indicated for the first-line treatment of adult patients with metastatic or with unresectable, recurrent head and neck squamous cell carcinoma (HNSCC) whose tumors express programmed death ligand 1 (PD-L1) [combined positive score (CPS) ≥1] as determined by an FDA-authorized test.
KEYTRUDA and KEYTRUDA QLEX™ (pembrolizumab and berahyaluronidase alfa-pmph) Injection 165 mg + 2,000 units/mL, as single agents, are each indicated for the treatment of adult patients with recurrent or metastatic head and neck squamous cell carcinoma (HNSCC) with disease progression on or after platinum-containing chemotherapy.